 Pharmaceutical Computer Systems Validation: Quality Assurance, Risk Management and Regulatory Compliance Second EDITION Other features of the Second Edition include: Case Study 20: Computer Applications Supporting the Supply of Biotechnology Products. Case Study 19: Process Analytical Technology. Case Study 18: Blood Establishment Computer Systems. Case Study 17: Medical Devices and Their Automated Manufacture. 
Case Study 16: Internet/Intranet Applications. Case Study 15: IT Infrastructure and Associated Services. Case Study 14: Marketing and Supply Applications. Case Study 13: Enterprise Resource Planning Systems. Case Study 12: Electronic Document Management Systems. Case Study 10: Desktop Applications Including Spreadsheets. Case Study 9: Engineering Management Systems. Case Study 8: Building Management Systems. Case Study 7: Manufacturing Execution Systems and Electronic Batch Records. Case Study 5: Control and Monitoring Instrumentation. Case Study 3: Laboratory Information Management Systems. Case Study 2: Chromatography Data Systems. Case Study 1: Computerized Analytical Laboratory Systems. 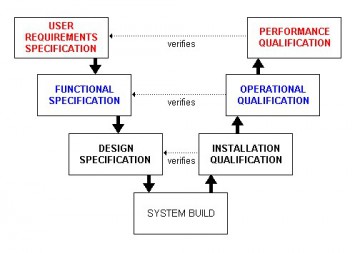
Electronic Records and Electronic Signatures. 
User Qualification and Authorization to Use. Requirements Capture and Supplier (Vendor)Selection. Project Initiation and Compliance Determination. Table of contents : Foreword to the Second Edition. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
